IN INDUSTRY-FUNDED STUDIES, TRUMP'S CHEMICAL SAFETY NOMINEE BACKED EXPOSURES 1,000 TIMES HIGHER THAN EPA’S RISK LEVEL
• EWG found 1,4-dioxane, a carcinogen, in nearly 90 million American’s drinking water supplies.
• 1,4-Dioxane was also found in thousands of personal care products.
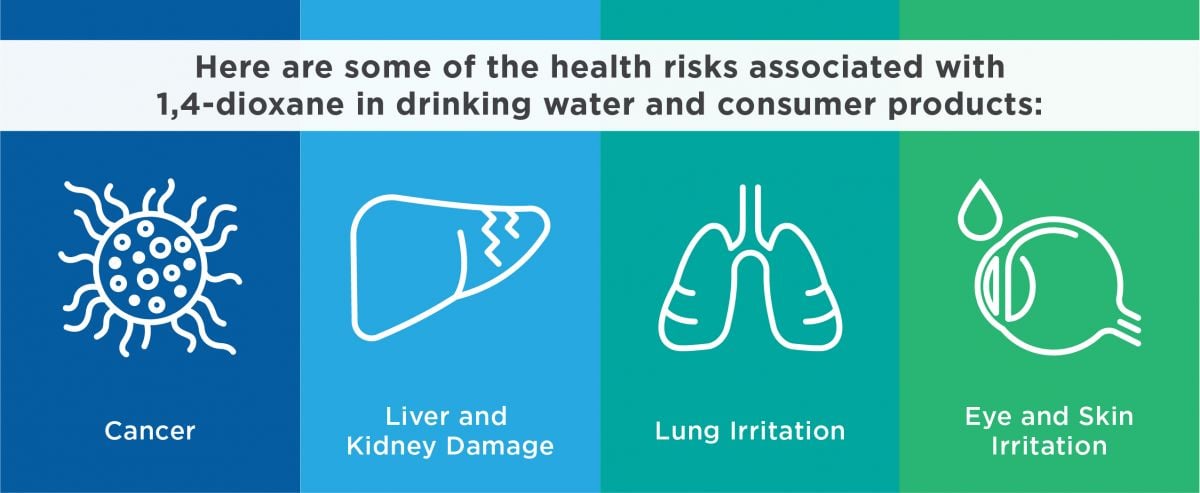
• Associated health risks include cancer, and liver and kidney damage.
• The contaminant is unregulated in both drinking water and cosmetics.
An industrial solvent classified as a likely carcinogen, which is also a common impurity in cosmetics and household cleaners, was detected in samples of drinking water supplies for nearly 90 million Americans in 45 states, according to testing data from local utilities analyzed by EWG.1
The tests showed that more than 7 million people in 27 states are served by public water systems with higher average levels of the chemical, 1,4-dioxane, than the level the Environmental Protection Agency considers to marginally increase cancer risk.2 That level, established by the EPA in 2013, is 0.35 parts per billion. EWG endorses this level as the no-compromise benchmark to fully protect public health.
But this health-based guideline does not set a legal limit for 1,4-dioxane in drinking water. The EPA has selected 1,4-dioxane as one of the first 10 chemicals to be reviewed under the nation’s new chemical safety law, but EWG is concerned, as President Trump’s nominee to head the EPA’s chemical safety office has authored two industry-funded studies arguing that people can safely be exposed to levels 1,000 times higher than the agency’s increased risk level for the chemical.
An interactive map shows test data for 1,4-dioxane in drinking water across the nation, with detailed information for each affected water system listed in EWG’s Tap Water Database. The database is the most comprehensive resource on U.S. drinking water available, aggregating and analyzing water quality information for nearly 50,000 public water systems in all 50 states.
From 2010 to 2015, 1,4-dioxane was detected in 1,060 public water systems, according to data from tests mandated by the EPA under the Unregulated Contaminant Monitoring Rule3 and additional tests conducted by water utilities themselves. Another 1,167 systems receive some or all of their treated water from utilities that detected 1,4-dioxane, spreading the contamination beyond the utility that originally found it.

The extent of nationwide contamination is likely greater than what is shown in the map and database. Small and medium-sized systems very rarely test for 1,4-dioxane, and private water wells – which draw on groundwater that can be contaminated by leaks from dump sites – are exempt from testing.
A closer look at the test data shows:
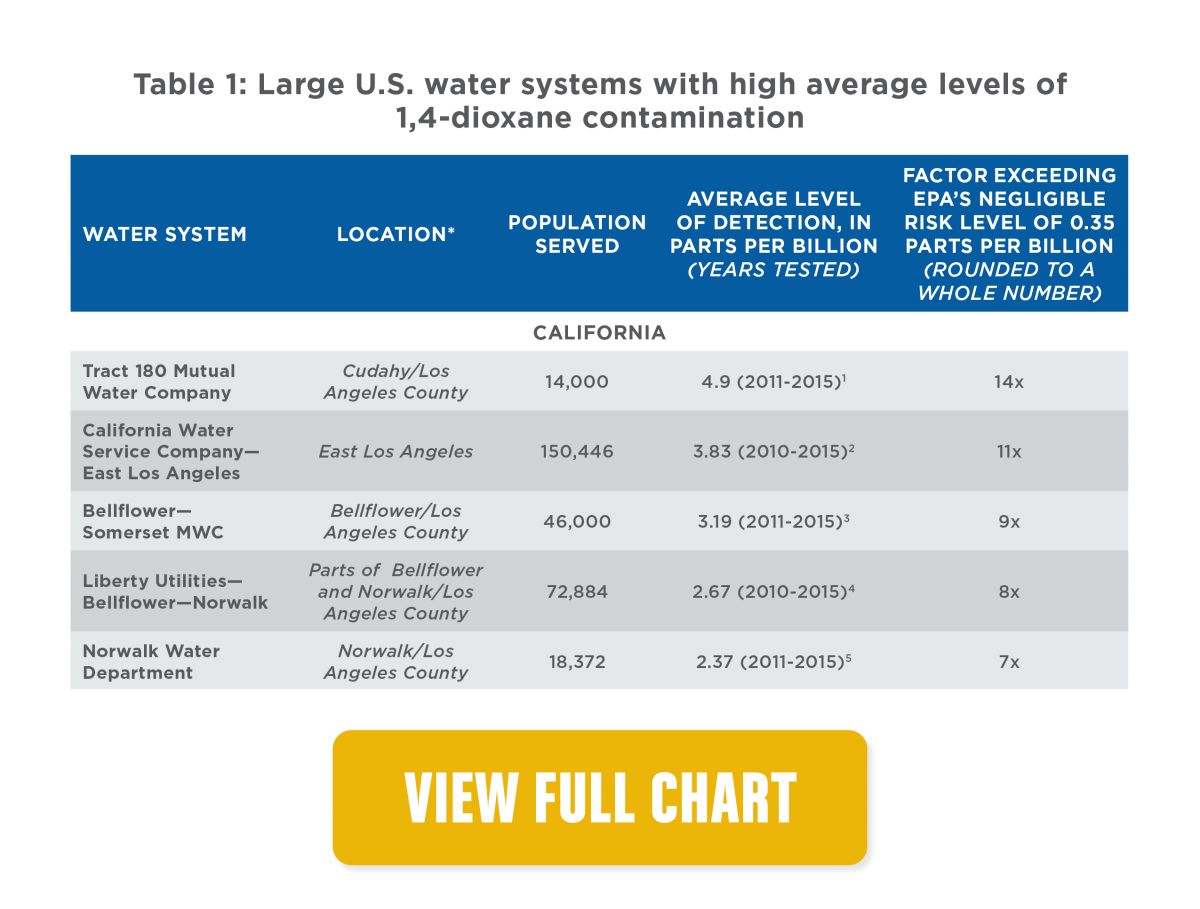
- Ranking the most-contaminated water systems, hot spots include the Cape Fear River basin in North Carolina, affecting Fayetteville and surrounding communities; southeastern Los Angeles County, Calif.; and New York’s Long Island. Samples from water systems serving those areas had average levels of 1,4-dioxane ranging from four times to about 17 times the EPA's increased cancer risk level (see Table 1 below).
- States with the most people exposed to 1,4-dioxane above the increased risk level are California, with 2.5 million people exposed; North Carolina, with 1.2 million; and New York, with 700,000.
- Some utilities have shut down wells contaminated with1,4-dioxane, limiting their community’s water supply. One well in the Hicksville Water District on Long Island had one of the highest levels of 1,4-dioxane detected in the nation – 33 parts per billion, more than 90 times the increased risk level – forcing its closure in 2016.4 In 2015, New Brighton, Minn., had to shut down six contaminated wells and switch to using water from Minneapolis.5
- 1,4-Dioxane in drinking water sources can come from wastewater discharges, toxic waste and Superfund sites, as well as industrial facilities where plastics and solvents have been manufactured or used.6 Tracing this contamination to a specific source can be difficult because 1,4-dioxane can be carried, through ground or surface water, away from the original discharge site.
An industrial solvent linked to cancer, liver and kidney damage
1,4-Dioxane has been used since the 1950s as an industrial solvent to dissolve greasy and oily substances, and as a chemical stabilizer.7 In 2015, approximately 1 million pounds of 1,4-dioxane were produced or imported into the U.S., as reported to the EPA, and 675,000 pounds were released into the environment. Two companies – BASF Corporation in Baton Rouge, La., and Hamilton, Ohio; and Tedia Company in Butler, Ohio – reported that they produced and imported it, and a third company that masked its name under a claim of confidentiality reported importing the chemical in 2015.8 1,4-Dioxane is also a byproduct of plastic production and other manufacturing processes.
The EPA has classified 1,4-dioxane as a likely human carcinogen,9 and it is listed in the State of California’s official registry of chemicals known to cause cancer.10 In animal studies, 1,4-dioxane exposure from drinking water caused tumors in the liver, nasal cavity, peritoneal and mammary glands.11
Short-term exposure to relatively high amounts of 1,4-dioxane is particularly damaging to the liver and kidneys.12 Some occupational studies of pregnant workers exposed to 1,4-dioxane and other solvents have reported higher rates of pregnancy loss, stillbirths, premature births and low birth weights. But it is unclear whether those effects can be attributed solely or in part to 1,4-dioxane exposure.13
As a result of both direct use and disposal, 1,4-dioxane often shows up in the wastewater that industrial facilities discharge to creeks, rivers and municipal sewer systems – a practice that has gone largely unchecked for decades. There is no federal regulation limiting 1,4-dioxane levels under the Safe Drinking Water Act. But the EPA Office of Research and Development has defined a concentration of 0.35 parts per billion, or ppb – about one drop of water in three Olympic-size swimming pools – as the amount of 1,4-dioxane expected to cause no more than one additional case of cancer in 1 million people who drink and bathe with the water over a lifetime.
1,4-Dioxane also found in consumer products
Drinking water is not the only source of 1,4-dioxane exposure for Americans.
The chemical is a common impurity in cosmetics and household cleaning products, which occurs as a result of a process called ethoxylation.14 Some products in which 1,4-dioxane contamination is found include shampoos, foaming soaps, bubble bath, lotions and laundry soap. In 2009, tests by the Campaign for Safe Cosmetics detected 1,4-dioxane in two-thirds of bath products tested.15 In August, Hong Kong’s Consumer Council tested 60 samples of shampoo, including products from brands sold in the U.S., and found 1,4-dioxane in 38 samples, or 63 percent. More than 8,000 products in EWG’s Skin Deep® cosmetics database include ingredients produced through ethoxylation.16
The International Cooperation on Cosmetics Regulation, a global group of regulatory authorities, has recommended phasing in a limit of 10 parts per million, or ppm, on 1,4-dioxane in cosmetics, but this limit is not mandatory.17 The Food and Drug Administration also says there’s a simple process, called vacuum stripping, for minimizing 1,4-dioxane in these products.18 But the lack of reporting and labeling requirements mean it is unclear how many companies take care to remove it.19
1,4-dioxane can also be present in paint strippers, dyes, greases, waxes and varnishes. Residues of 1,4-dioxane are sometimes found in food additives, food packaging and produce sprayed with pesticides that contain the chemical.20 The FDA has set a limit of 10 ppm for 1,4-dioxane in glycerides and polyglycerides used in hydrogenated vegetable oils.21
Because of its wide use and potential harm, 1,4-dioxane is one of the first 10 chemicals the EPA picked for review under the nation’s new chemical safety law. But in risk evaluation documents released in June, the EPA indicated it will leave critical exposures to 1,4-dioxane out of its assessment , including consumer exposures through personal care and cleaning products. This faulty approach would likely result in a dramatic underestimate of 1,4-dioxane exposures Americans face.
In industry-funded studies, key EPA nominee downplayed dangers
Michael Dourson, President Trump’s nominee to lead the EPA’s chemicals and pesticides office, is the founder and head of TERA, a consulting firm that has consistently worked to undercut chemical regulation.
Dourson authored two papers released in 2014 and 2017, arguing that people can safely be exposed to 1,4-dioxane levels of 350 ppb – 1,000 times greater than the EPA’s increased cancer risk level.22 This work was funded by PPG Industries, a manufacturer of paints and coatings in Circleville, Ohio. 1,4-Dioxane released from the company’s factory has traveled underground in a plume of contamination, affecting the Earnhart Hill Water District public water supply.23
The thousand-fold difference between Dourson’s recommendation and the EPA’s increased cancer risk level24 is based on his assertion that 1,4-dioxane cannot initiate liver cancer at low concentrations, a statement that contradicts the weight of scientific evidence. Table 2 summarizes the contrast between the health-protective approach taken by the EPA and Dourson’s assumptions.
In 2015, scientists at the Michigan Department of Environmental Quality reviewed the arguments presented by Dourson and said that the “available scientific information regarding the carcinogenicity of 1,4-dioxane does not support TERA’s hypothesis and are insufficient to deviate” from the EPA’s assumptions used to calculate the potential for cancer risk.25 The scientists added that the “most compelling argument” for following the EPA’s assumptions was the presence of multiple types of tumors in exposed test animals, “all of which are relevant to humans.”
Dourson’s industry-funded work to downplay the dangers of 1,4-dioxane is a red-flag reason he should not head the EPA’s chemical and pesticide safety office. Entrusting him with pending decisions about 1,4-dioxane regulation would endanger the health of people living near contaminated sites, those whose water supplies are tainted with this cancer-causing chemical, and everyone who uses products that may contain 1,4-dioxane as an impurity.
The widespread contamination of drinking water with 1,4-dioxane makes it imperative for the EPA to go beyond the non-enforceable increased cancer risk guideline by setting an enforceable standard, known as a maximum contaminant level, or MCL. The EPA should also take action to minimize 1,4-dioxane releases into waterways.
It’s equally important to address contamination at its source, keeping 1,4-dioxane from polluting surface waters that supply drinking water. Industries can reduce releases of 1,4-dioxane by modifying their manufacturing practices, using different raw materials, treating wastes and through other measures. 1,4-Dioxane released from municipal wastewater dischargers can be a significant source of contamination for utilities and must be limited. More stringent state and federal regulations for 1,4-dioxane releases into rivers and streams are essential, because these discharges contaminate drinking water sources for millions of Americans.

The federal government could take several important steps to protect consumers from 1,4-dioxane. In particular, the EPA has the authority to set a legal limit for 1,4-dioxane in drinking water and regulate industrial uses of 1,4-dioxane under the Toxic Substances Control Act, and the FDA has the authority to regulate 1,4-dioxane in personal care products like shampoos.
In November 2016, the EPA selected 1,4-dioxane as one of the first 10 chemicals it would review under the new chemical safety law passed earlier that year. As part of this review, the EPA must look at all potential uses and exposures to 1,4-dioxane, including chemical manufacturing and processing, contaminated personal care and cleaning products, water contamination, accidental spills and disposal processes. The EPA must also take action as part of this review to limit all potential exposures to 1,4-dioxane by setting health-protective restrictions on its use.
But risk evaluation documents released in June did not propose limiting or even considering all 1,4-dioxane exposures. Furthermore, the proposed scope of the EPA’s 1,4-dioxane assessment leaves out important uses and exposures, including likely consumer exposures through personal care and cleaning products.
The FDA can also take action to ensure 1,4-dioxane is removed or virtually eliminated from personal care products such as shampoos, shower gels, body washes, foaming hand soaps, bubble baths and lotions. The FDA has a recommended process for vacuum-stripping 1,4-dioxane from these products and has the authority to require companies to do so. Manufacturers also can use additional controls to slow the formation of 1,4-dioxane as a byproduct, leaving much smaller amounts in finished products. Using these controls reduces the amount of 1,4-dioxane formed purposely, lowering worker exposures and the amount of 1,4-dioxane waste.
Sens. Charles Schumer and Kirsten Gillibrand, both of New York, recently sent a joint petition to the FDA asking the agency to ban 1,4-dioxane in personal care products.26
“Because 1,4-dioxane can cause cancer, serves no purpose in cosmetics, is not identified on packaging, and can be minimized during production, FDA should protect the public by requiring all companies to utilize the technologies necessary to limit its presence to below detectable levels,” Schumer said.
The FDA should approve this petition and regulate 1,4-dioxane in personal care products. Congress should also increase the FDA’s limited authority over personal care products so that the agency can do more to address contaminants like 1,4-dioxane and other dangerous chemicals.
However, cutting funding for the EPA and the FDA, as the Trump administration has proposed, and subjecting new agency rules to needless delays will make it far more difficult for these agencies to protect consumers from 1,4-dioxane. Congress should reject proposals to cut the EPA’s and the FDA’s funding, and should reject bills like the Regulatory Accountability Act, which would erect needless hurdles for agencies trying to adopt critical consumer protections.
In the absence of federal regulation, some states have set water quality limits, guidelines and notification requirements related to the chemical:
- California: Water utilities must notify state government agencies whenever 1,4-dioxane is detected at concentrations above 1 ppb.27
- Colorado: 1,4-Dioxane levels in groundwater should not exceed 0.35 ppb.28
- Maine: The drinking water maximum exposure guideline is 4 ppb.29
- Massachusetts: The non-enforceable drinking water guideline is 0.3 ppb.30
- New Hampshire: The reporting limit for public water supplies is 0.25 ppb.31
- New Jersey: The groundwater quality standard is 0.4 ppb.32
- North Carolina: The groundwater quality standard is 3 ppb and the surface water supply standard is 0.35 ppb.33
More states should not wait for the federal government, but move forward to set their own health-protective limits and guidelines for 1,4-dioxane in drinking water and groundwater. States that have only have non-enforceable guidelines should instead create legal limits.
How to protect yourself and your family from 1,4-dioxane
Until the federal and state governments set tough limits on 1,4-dioxane, limiting exposure from all sources is the best thing one can do to protect health. Consumers can take several steps to reduce their exposures.
1,4-Dioxane contamination in drinking water is difficult to address with common home water treatment techniques. Carbon filters are ineffective, and while some reverse osmosis systems can remove a significant portion of 1,4-dioxane, they are not entirely effective. Reducing 1,4-dioxane levels in tap water can require additional, often costly, treatment technologies that many water utilities lack.34
1,4-Dioxane does not readily break down, so once it is in water it stays there unless removed.35 Americans living in areas with 1,4-dioxane contamination should call their water utilities to urge installation of specialized treatments to reduce levels of the chemical. Residents should also contact local and state elected officials and regulatory agencies to advocate for source water protection to prevent 1,4-dioxane contamination in the first place.
Citizens should encourage the companies that make personal care products to remove 1,4-dioxane from all potentially contaminated products. You can also avoid 1,4-dioxane by skipping products that contain PEG, polyethylene, polyethylene glycol, polyoxyethylene, polyoxynolethylene and chemicals ending in –eth and –oxynol.
Finally, citizens can tell their elected representatives to ensure that the EPA, the FDA and other federal agencies have sufficient resources and authority to do the important work of studying and regulating chemicals like 1,4-dioxane.
[1] EWG’s Tap Water Database. 1,4-Dioxane. Accessed Aug. 3, 2017. Available at www.ewg.org/tapwater/contaminant.php?contamcode=2049
[2] U.S. EPA Integrated Risk Information System, 1,4-Dioxane. 2013. Available at cfpub.epa.gov/ncea/iris2/chemicalLanding.cfm?substance_nmbr=326
[3] U.S. EPA, Occurrence Data for the Unregulated Contaminant Monitoring Rule. Last visited May 6, 2017. www.epa.gov/dwucmr/occurrence-data-unregulated-contaminant-monitoring-rule#3
[4] Staff report, Glass Half Empty. Hicksville News, March 16, 2017. Available at http://hicksvillenews.com/2017/03/16/glass-half-empty/
[5] Shannon Prather, Two North Metro Cities Scramble to Upgrade Water Plants to Filter Out Emerging Contaminant. Star Tribune, June 20, 2016. Available at http://www.startribune.com/two-north-cities-scramble-to-upgrade-water-plants-to-filter-out-emerging-contaminant/383693321/
[6] Water Research Foundation, 1,4-Dioxane White Paper. 2014. Available at www.waterrf.org/resources/StateOfTheScienceReports/1,4-dioxane.pdf
[7] Ibid.
[8] U.S. EPA Scope of the Risk Evaluation for 1,4-Dioxane, June 2017. Accessed Aug. 15, 2017. Available at www.epa.gov/sites/production/files/2017-06/documents/dioxane_scope_06-22-2017.pdf and U.S. EPA Chemical Data Reporting 2016 Submissions Database. Accessed Aug. 15, 2017. Available at java.epa.gov/chemview
[9] U.S. EPA Integrated Risk Information System, 1,4-Dioxane. 2013. Available at cfpub.epa.gov/ncea/iris2/chemicalLanding.cfm?substance_nmbr=326
[10] California Office of Environmental Health Hazard Assessment, Proposition 65 List. Accessed May 10, 2017. Available at oehha.ca.gov/proposition-65/chemicals/14-dioxane
[11] U.S. EPA, Technical Fact Sheet – 1,4-Dioxane. 2014, EPA 505-F-14-011. Available at www.epa.gov/fedfac/technical-fact-sheet-14-dioxane
[12] Agency for Toxic Substances & Disease Registry, Toxicological Profile for 1,4-Dioxane, Chapter 2: Relevance to Public Health. 2012. Available at www.atsdr.cdc.gov/toxprofiles/tp187-c2.pdf
[13] Agency for Toxic Substances & Disease Registry, Toxicological Profile for 1,4-Dioxane, Chapter 2: Relevance to Public Health. 2012. Available at www.atsdr.cdc.gov/toxprofiles/tp187-c2.pdf
[14] Agency for Toxic Substances & Disease Registry (ATSDR), Toxicological Profile for 1,4-Dioxane. 2012. Available at www.atsdr.cdc.gov/toxprofiles/tp.asp?id=955&tid=199
[15] Campaign for Safe Cosmetics, No More Toxic Tub: Getting Contaminants Out of Children’s Bath & Personal Care Products. 2009. Available at static.ewg.org/reports/2009/Campaign-for-Safe-Cosmetics-Report-No-More-Toxic-Tub.pdf?_ga=2.33873377.1534215448.1492697043-81690461.1469647247.
[16] Environmental Working Group, EWG Surveys Personal Care Product Companies About 1,4-Dioxane. 2017. Available at www.ewg.org/release/ewg-surveys-personal-care-product-companies-about-14-dioxane#.WZYctnd95TZ
[17] International Cooperation on Cosmetics Regulation, Considerations on Acceptable Trace Level of 1,4-Dioxane in Cosmetic Products. 2017. Available at www.iccrnet.org/files/2414/8717/1555/ICCR_14-Dioxane_Final_2017.pdf
[18] U.S. FDA, 1,4-Dioxane A Manufacturing Byproduct. Last visited May 6, 2017. Available at www.fda.gov/Cosmetics/ProductsIngredients/PotentialContaminants/ucm101566.htm
[19] Environmental Working Group, EWG Research Shows 22 Percent of All Cosmetics May Be Contaminated with Cancer-Causing Impurity. 2007. Available at www.ewg.org/news/news-releases/2007/02/08/ewg-research-shows-22-percent-all-cosmetics-may-be-contaminated-cancer
[20] Water Research Foundation, 1,4-Dioxane White Paper. 2014. Available at www.waterrf.org/resources/StateOfTheScienceReports/1,4-dioxane.pdf
[21] 21 C.F.R. § 172.736. Available at www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfCFR/CFRSearch.cfm?fr=172.736
[22] Michael Dourson et al., Mode of Action Analysis for Liver Tumors from Oral 1,4-Dioxane Exposures and Evidence-Based Dose Response Assessment. Regulatory Toxicology and Pharmacology, April 2014. Available at www.sciencedirect.com/science/article/pii/S0273230014000129?via%3Dihub
and Michael Dourson et al., Update: Mode of Action (MOA) for Liver Tumors Induced by Oral Exposure to 1,4-Dioxane. Regulatory Toxicology and Pharmacology, August 2017. Available at www.sciencedirect.com/science/article/pii/S0273230017300429
[23] Ohio Environmental Protection Agency, Environmental Response and Revitalization site summary PPG Industries, Inc. 2012. Accessed Aug. 3, 2017. Available at www.epa.ohio.gov/cdo/ppg.aspx
[24] U.S. EPA Integrated Risk Information System, 1,4-Dioxane. 2013. Available at cfpub.epa.gov/ncea/iris2/chemicalLanding.cfm?substance_nmbr=326
[25] Michigan Department of Environmental Quality, Review of a 1,4-Dioxane Presentation by Michael Dourson, Ph.D., on Oct. 8, 2013. February 2015. Available at www.michigan.gov/documents/deq/deq-aqd-toxics-14-DioxaneTSG_Report_2015_487415_7.pdf
[26] U.S. Sen. Charles E. Schumer, Press Release, April 13, 2017. Available at www.schumer.senate.gov/newsroom/press-releases/schumer-14-dioxane-likely-carcinogen-in-li-water-supply-emanates-from-consumer-and-kids-products-like-shampoo_hand-soap-senator-calls-on-fda-to-require-companies-to-finally-strip-dangerous-toxin-action-will-make-products--li-water-supply-safer-for-countless-families
[27] California State Water Resources Control Board, Drinking Water Notification Levels. 2015. Available at www.waterboards.ca.gov/drinking_water/certlic/drinkingwater/documents/notificationlevels/notificationlevels.pdf
[28] Colorado Department of Public Health and the Environment, Regulation No. 41: The Basic Standards for Ground Water (5 CCR 1002-41); Water Quality Control Commission. 2012. Available at www.colorado.gov/pacific/sites/default/files/41_2016%2812%29.pdf
[29] Maine Department of Human Services, Maximum Exposure Guidelines (MEGs) for Drinking Water, 2016. Available at www.maine.gov/dhhs/mecdc/environmental-health/eohp/wells/documents/megtable2016.pdf
[30] Massachusetts Department of Environmental Protection, Standards and Guidelines for Contaminants in Massachusetts Drinking Waters. 2014. Available at www.mass.gov/eea/agencies/massdep/water/drinking/standards/standards-and-guidelines-for-drinking-water-contaminants.html#Guidelines
[31] New Hampshire Department of Environmental Services, Change in Reporting Limit for 1,4-Dioxane. 2011. Available at www.des.nh.gov/organization/divisions/waste/hwrb/sss/hwrp/documents/report-limits14dioxane.pdf
[32] New Jersey Department of Environmental Protection, Ground Water Quality Standard 1,4-Dioxane. 2015. Available at www.nj.gov/dep/wms/bears/docs/1,4%20dioxane%20final%20draft%20for%20posting2.pdf
[33] North Carolina’s surface water supply standard is unique in that it is a calculated standard. It is based on 15NCAC 02B .0208 Standards for Toxic Substances. For carcinogens, this concentration is based on the one-in-a-million cancer risk level (0.35 ppb from EPA IRIS 2013). If this standard for cancer risk changes over time, so will the water supply standard. See: North Carolina Department of Environmental Quality, North Carolina’s Surface Water Quality Standards. Available at www.deq.nc.gov/document/nc-stds-groundwater-02lstandards and www.reports.oah.state.nc.us/ncac/title%2015a%20-%20environmental%20quality/chapter%2002%20-%20environmental%20management/subchapter%20b/subchapter%20b%20rules.pdf
[34] Water Research Foundation, 1,4-Dioxane White Paper. 2014. Available at www.waterrf.org/resources/StateOfTheScienceReports/1,4-dioxane.pdf
[35] Minnesota Department of Health, Environmental Health Division, 1,4-Dioxane in Drinking Water. 2015. Available at www.health.state.mn.us/divs/eh/risk/guidance/dwec/dioxaneinfo.pdf